
Xylene
Organic compounds with the formula (CH3)2C6H4 / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Xylene?
Summarize this article for a 10 year old
In organic chemistry, xylene or xylol (from Greek ξύλον (xylon) 'wood';[1][2] IUPAC name: dimethylbenzene) are any of three organic compounds with the formula (CH3)2C6H4. They are derived from the substitution of two hydrogen atoms with methyl groups in a benzene ring; which hydrogens are substituted determines which of three structural isomers results. It is a colorless, flammable, slightly greasy liquid of great industrial value.[3]
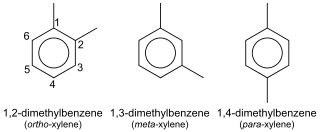
The mixture is referred to as both xylene and, more precisely, xylenes. Mixed xylenes refers to a mixture of the xylenes plus ethylbenzene. The four compounds have identical molecular formulas C8H10. Typically the four compounds are produced together by various catalytic reforming and pyrolysis methods.[4]