
Cannabidiol
Phytocannabinoid discovered in 1940 / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Cannabidiol?
Summarize this article for a 10 year old
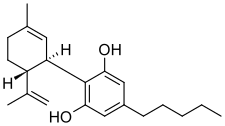
Cannabidiol (CBD) (/kæ.nə.bə.ˈdaɪ.əl/) is a phytocannabinoid discovered in 1940. It is one of 113 identified cannabinoids in cannabis plants, along with tetrahydrocannabinol (THC), and accounts for up to 40% of the plant's extract.[17] As of 2022[update], clinical research on CBD included studies related to the treatment of anxiety, addiction, psychosis, movement disorders, and pain, but there is insufficient high-quality evidence that cannabidiol is effective for these conditions.[18][19][20][21] CBD is also sold as a herbal dietary supplement promoted with unproven claims of particular therapeutic effects.[22]
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Epidiolex, Epidyolex |
| Other names | CBD, cannabidiolum, (−)-cannabidiol[1] |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a618051 |
| License data |
|
| Pregnancy category |
|
| Addiction liability | Low[3] |
| Routes of administration | Inhalation (smoking, vaping), buccal (aerosol spray),[4][5] oral (solution)[6][7] |
| Drug class | cannabinoid |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | |
| Elimination half-life | 18–32 hours[15] |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank |
|
| ChemSpider |
|
| UNII | |
| KEGG | |
| ChEBI | |
| PDB ligand | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.215.986 |
| Chemical and physical data | |
| Formula | C21H30O2 |
| Molar mass | 314.469 g·mol−1 |
| 3D model (JSmol) | |
| Melting point | 66 °C (151 °F) |
| Boiling point | 160–180 °C (320–356 °F) [16][unreliable medical source?] |
| Solubility in water | Insoluble |
| |
| |
| (verify) | |
Cannabidiol can be taken internally in multiple ways, including by inhaling cannabis smoke or vapor, oral, and as an aerosol spray into the cheek.[4][5] It may be supplied as CBD oil containing only CBD as the active ingredient (excluding THC or terpenes), CBD-dominant hemp extract oil, capsules, dried cannabis, or prescription liquid solution.[7][20] CBD does not have the same psychoactivity as THC,[23][24] and can modulate the psychoactive effects of THC on the body if both are present.[17][23][25][26] CBD heated to 250–300 °C may partially be converted into THC.[27]
In the United States, the cannabidiol drug Epidiolex was approved by the Food and Drug Administration (FDA) in 2018 for the treatment of two epilepsy disorders.[7] While the 2018 United States Farm Bill removed hemp and hemp extracts (including CBD) from the Controlled Substances Act, the marketing and sale of CBD formulations for medical use or as an ingredient in dietary supplements or manufactured foods remains illegal under FDA regulation, as of 2024[update].[28][29]