
Tris(2-phenylpyridine)iridium
Chemical compound / From Wikipedia, the free encyclopedia
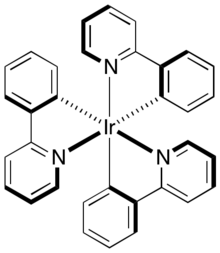
Tris(2-phenylpyridine)iridium, abbreviated [Ir(ppy)3] is the organoiridium complex with the formula Ir(C6H4-C5H4N)3. The complex, a yellow-green solid, is a derivative of Ir3+ bound to three monoanionic 2-pyridinylphenyl ligands. It is electroluminescent, emitting green light. The complex is observed with the facial stereochemistry, which is chiral.
Quick Facts Names, Identifiers ...
 | |
| Names | |
|---|---|
| Other names
tris(2-phenylpyridine)iridium, Tris[2-(2-pyridinyl)phenyl]iridium, Tris[2-(2-pyridinyl)phenyl-C,N]iridium | |
| Identifiers | |
| ChemSpider | |
| ECHA InfoCard | 100.163.509 |
PubChem CID |
|
CompTox Dashboard (EPA) |
|
| |
| Properties | |
| C33H24IrN3 | |
| Molar mass | 654.793 g·mol−1 |
| Appearance | yellow-green solid |
| Hazards | |
| GHS labelling: | |
 | |
| Warning | |
| H315, H319, H335 | |
| P261, P264, P271, P280, P302+P352, P304+P340, P305+P351+P338, P312, P321, P332+P313, P337+P313, P362, P403+P233, P405, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close
The complex is prepared by cyclometalation reactions of 2-phenylpyridine and iridium trichloride, as represented by this idealized equation:[1][2]
- IrCl3 + 3 C6H5-C5H4N → Ir(C6H4-C5H4N)3 + 3 HCl
The complex and many analogues have been investigated for application in photoredox catalysis. Its excited state has a reduction potential of −2.14 V, nearly 1 V more negative than the reduction potential of excited [Ru(bipy)3]2+.[3]